T dm1 adjuvant3/28/2023 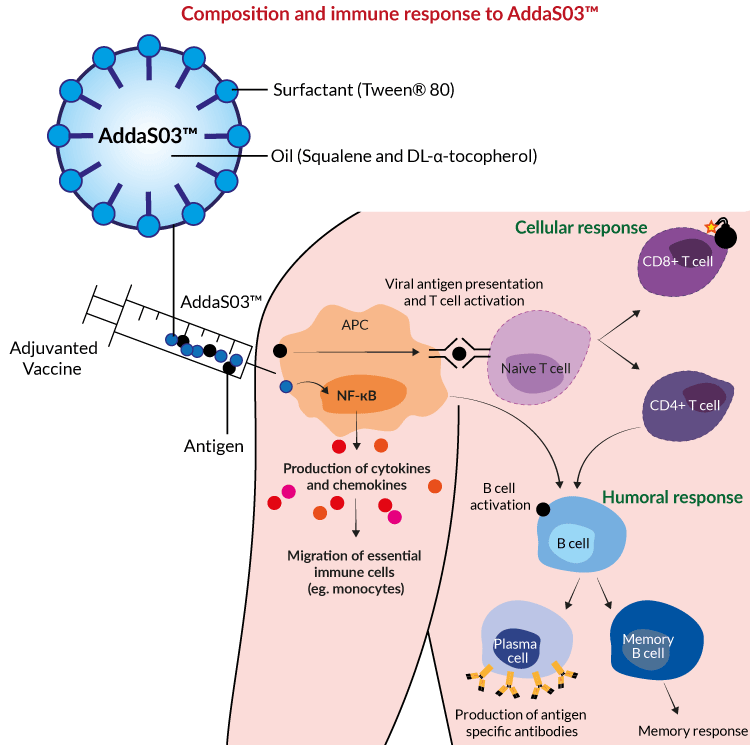
Adverse events (AEs) reported in previous studies to have occurred more commonly with T-DM1 or trastuzumab (eg, hepatotoxicity, peripheral neuropathy, hemorrhage, infusion-related reaction/hypersensitivity, and cardiac dysfunction) had similar all-grade incidence rates between the AC- and non-AC cohorts.  
Overall, the safety profile was similar between the AC and non-AC arms. These data compare favorably with the overall population results of the KATHERINE trial, in which T-DM1 reduced the risk of invasive disease recurrence or death by 50% (HR, 0.50 95% CI, 0.39-0.64 P 12 weeks. In the non-AC arm, the 3-year iDFS rates were 91.7% versus 81.4%, respectively. In the AC arm, the 3-year iDFS rates were 87.4% with T-DM1 versus 75.7% with trastuzumab. Mano, MD, PhD, presented a poster with several subanalyses of the trial, including a comparison of patients who received anthracycline (AC)-based NACT versus those who received non-AC-based NACT. Prior neoadjuvant therapy had to consist of ≥6 cycles of chemotherapy containing a taxane (with or without anthracycline) and ≥9 weeks of trastuzumab.Īt the 2019 San Antonio Breast Cancer Symposium (SABCS), lead study author Max S. The KATHERINE trial included 1486 patients with HER2-positive, nonmetastatic invasive primary breast cancer who were randomized in a 1:1 ratio to adjuvant T-DM1 or trastuzumab.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
